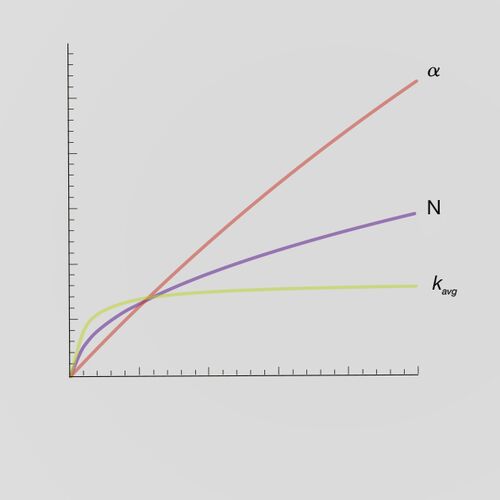
Important Considerations for GLP-1 Agonist Purification
-
A purification approach via RP is the ideal strategy
-
A screening of multiple selectivies leads to optimal results
-
Elevated pH is a Must-Have for an efficient purification
-
Regular alkaline CIP procedures improve the column lifetime
-
Highly robust stationary phases improve the productivity
- Reliable Supply and Regulatory Support is a Must for Save Production Processes

Innovation for all applications
High Performance Products for Purification and Analysis: YMC-Triart
HPLC/YMC-Triart-Symbol.jpg)
To overcome the limitations of traditional silica-based stationary phases regarding the chemical stability, hybrid silica-based phases have been developed. Hybrid silica contains organic groups within the silicon dioxide network, making it more resistant to a wider pH range
The innovative RP stationary phase YMC-Triart combines mechanical strength and pH stability. The stationary phases are availbe in analytical partical sizes with 1,9 - 5 µm (YMC-Triart (U)HPLC columns) andnlarger particle sizes for effeicient purification processes (YMC-Triart Prep). with full scalability.
Available Products
YMC-Triart RP Materials: Advantages for Purification and Analysis
- Versatility
- high chemical stability
- Method flexibility
- Robustness and Reproducibility
- alkaline CIP stability
- Full scalability from analytical to prep scale
- Low column back-pressure
- Improved loadability
Whitepaper for
Strategic peptide purification

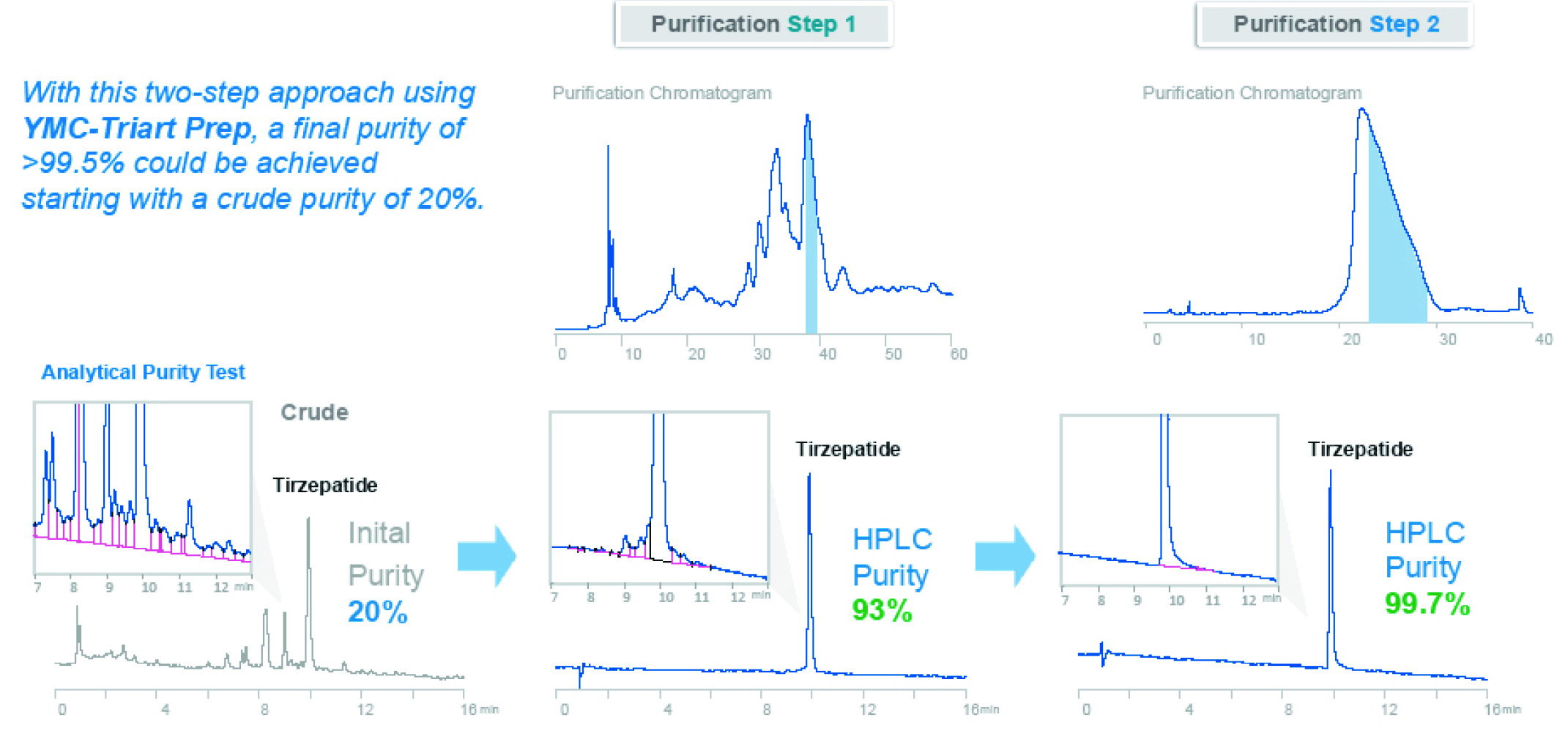
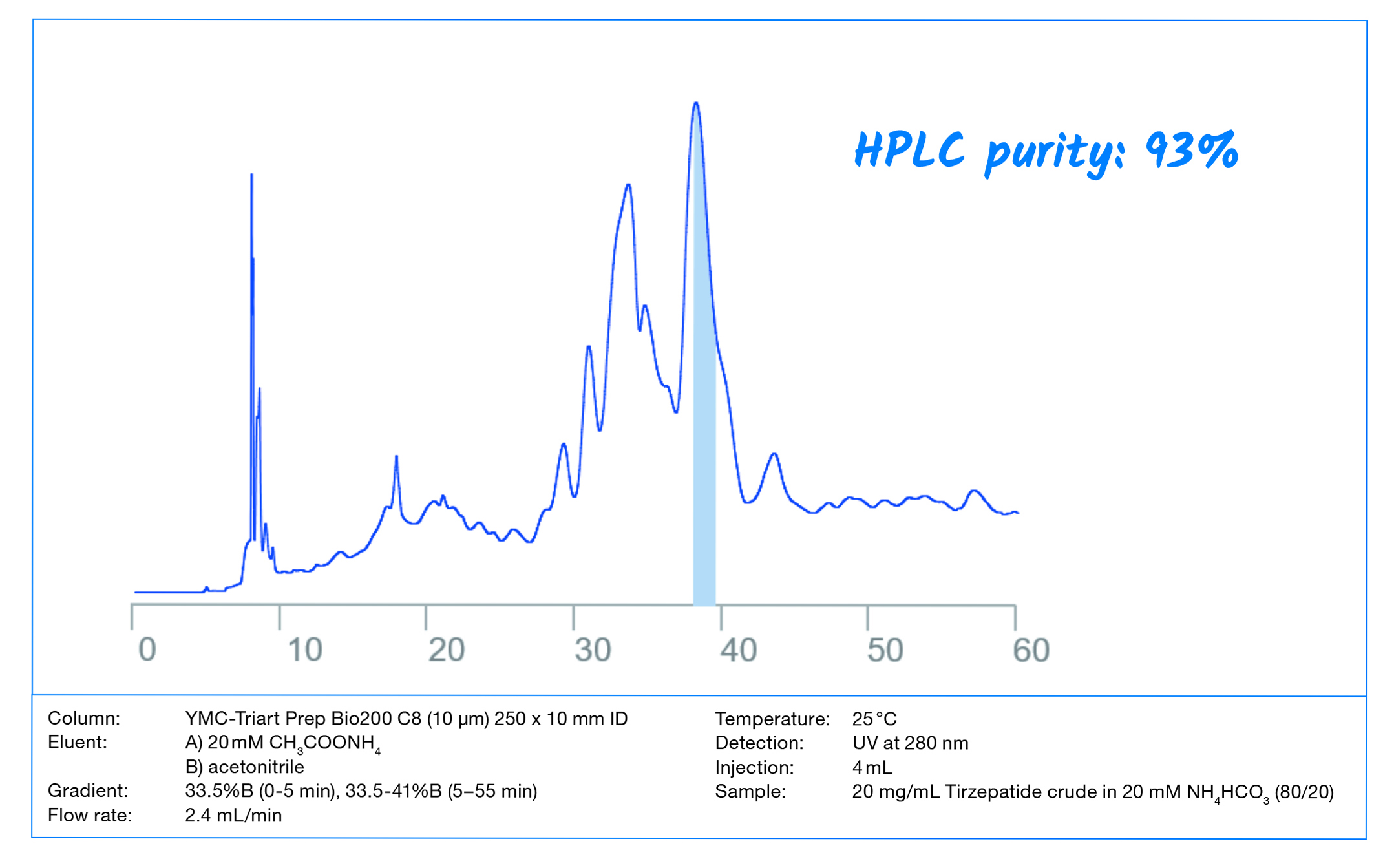
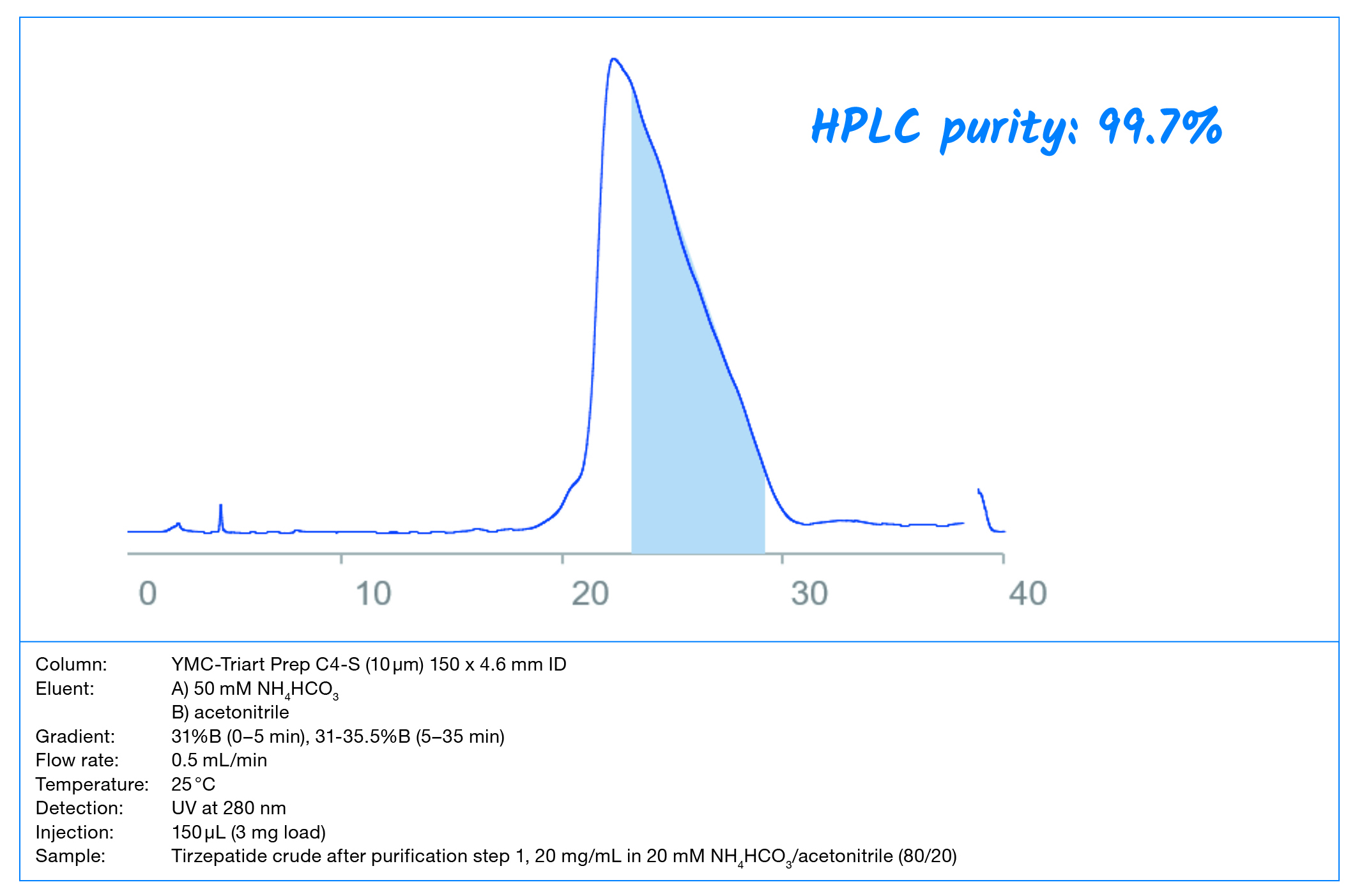
Peptides have become one of the most important biopharmaceuticals. This document is a guide for developing preparative peptide purification strategies with the considerations of economic and productive aspects. This means: maximum sample recovery in the shortest cycle time within cost limitations.
Contents of the Whitepaper
Within this Whitepaper, the important steps in method development for
peptide purifications using RP chromatography are demonstrated on a real-life example:
- Phase selection
- Pore size
- Particle size
- Mobile phase composition
- Gradient optimisation
Deepen your Knowledge
Seminar on GLP-1 Agonist Purificaiton

Meet our experts and gain comprehensive and clear knowledge about the purification of GLP-1 Agonists. This is a great opportunity to deepen your understanding and gather valuable insights.
| Title | Document Type | Language | Download |
 YMC-Triart Prep Packing Materials YMC-Triart Prep Packing Materials |
Brochure | EN | |
 YMC-Triart Prep Care and Use YMC-Triart Prep Care and Use |
Care and Use Instructions | EN | |
 Selection Tool for the YMC-Triart Prep Family - First Choice in Preparative LC Selection Tool for the YMC-Triart Prep Family - First Choice in Preparative LC |
Flyer | EN | |
 Packing Silica and Hybrid-Silica Stationary Phases into DAC Columns Packing Silica and Hybrid-Silica Stationary Phases into DAC Columns |
Overview | EN | |
 YMC Bulk Packing Materials for RP-, NP- and Chiral Prep. LC YMC Bulk Packing Materials for RP-, NP- and Chiral Prep. LC |
Overview | EN | |
 YMC Resins for Preparative Bioseparations YMC Resins for Preparative Bioseparations |
Overview | EN | |
 Purification method development for Liraglutide Purification method development for Liraglutide |
Poster | EN | |
 Loadability and productivity for prep LC processes Loadability and productivity for prep LC processes |
Technical Note | EN | |
 Packing Properties of YMC-Triart Prep Packing Properties of YMC-Triart Prep |
Technical Note | EN | |
 Sustainability and Mechanical Stability of YMC-Triart Prep Sustainability and Mechanical Stability of YMC-Triart Prep |
Technical Note | EN |
Expertise portal: More expert content for you
In our expertise area, we have compiled extensive knowledge and supporting digital services for you. Use our extensive knowledge database and the numerous calculators of our expertise portal.
Online presentations: Experience us live
In our online presentations, we pass on our expert knowledge to you and answer all your questions. Book an online presentation on a specific topic and meet us live.
Downloads: Our expert library
Our download area offers a wide range of knowledge about our products. Discover our extensive expert database with many informative downloads.